Towards Solving Brain Aging
The most important problem in medicine, and the case for acting now.
Neurodegenerative disease is the slow erasure of what we have most reason to value. Among the major diseases of aging, it is the one that takes the person before it takes the body. Pulling effective therapy forward by even a year is a year of selfhood preserved that would otherwise have been lost. Yet the field has converged on a narrow set of bets, like protein aggregate hypotheses, animal models that compress decades into months, and neuron-first thinking, the result of which has been the highest attrition rate in medicine. We think the way out is structural: attack brain aging where it is most tractable today, and build the models, tools, and platforms the field will need to attack it everywhere else tomorrow.
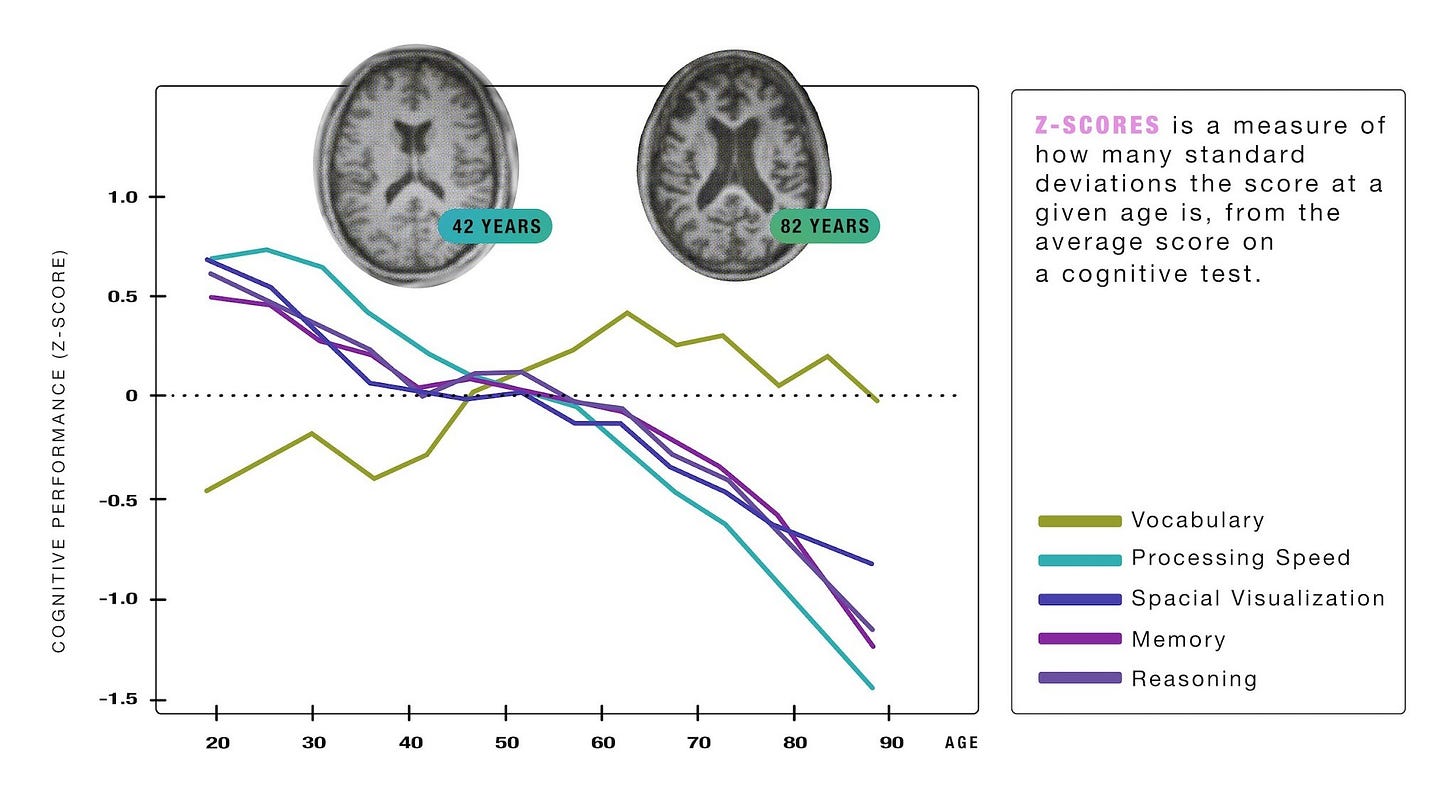
Brain aging is associated with broad-based declines in cognitive function, even in the absence of overt disease. From our Bottlenecks of Aging publication.
The ultimate solution to neurodegeneration will tackle the brain-aging biology that precedes it directly, or perhaps will even involve replacing brain tissue wholesale. But there is still much we don’t know about the biology, replacement technologies are many years away, and patients alive today cannot wait. The HIV epidemic offers the precedent: the cohort that survived past the 1996 introduction of HAART has gone on to long-acting prophylaxis, gene-edit trials, and the prospect of functional cure. Time bought by the first wave of therapies became time enough for the second. Our thinking mirrors that two-horizon strategy. Below we outline our thinking on neurodegeneration, with examples drawn from companies we've backed.
Modifying the disease in flight
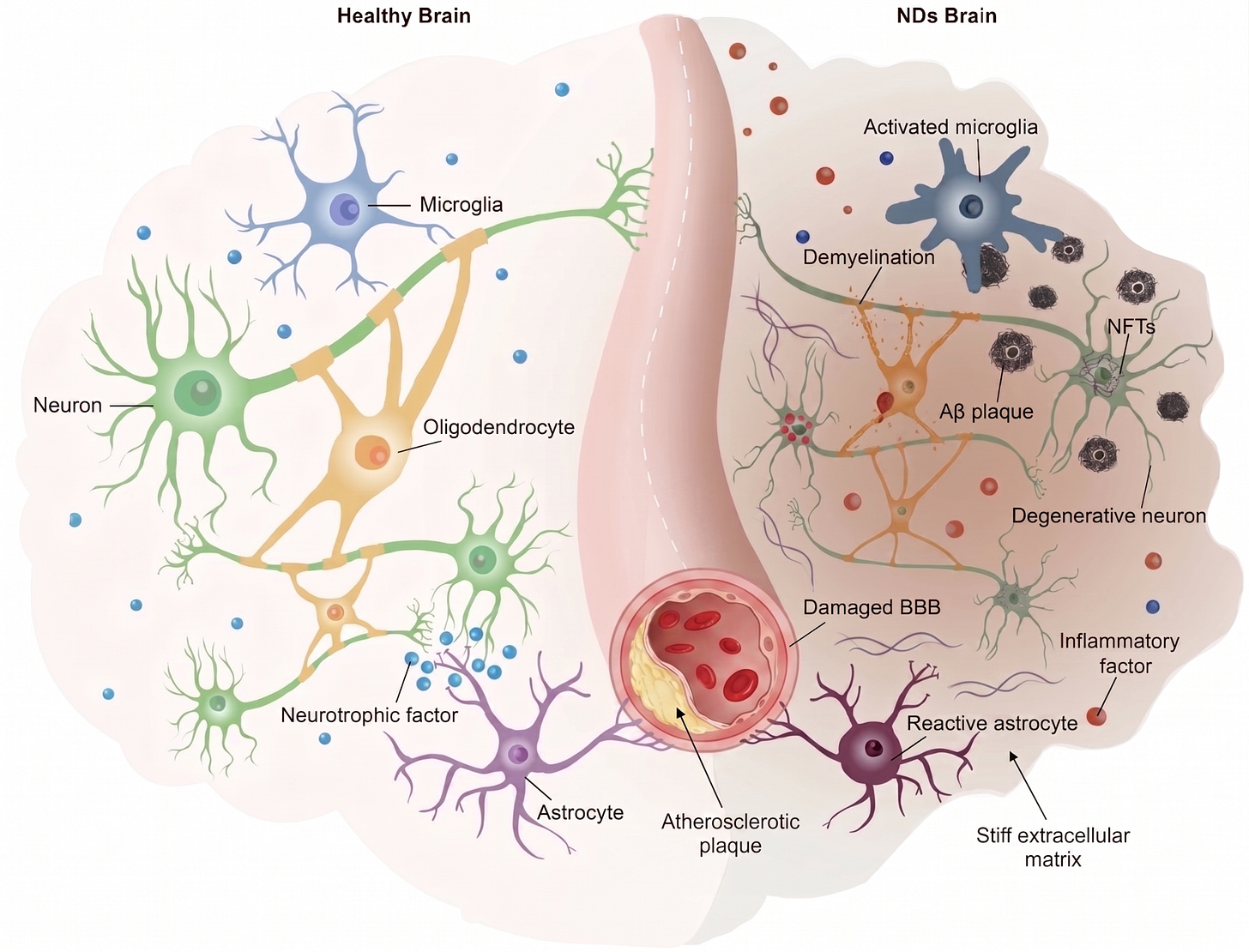
Neurodegenerative disorders display a common set of contributing factors, some of which are tractable today. Adapted from Jiang et al. (2025).
In the short term, we focus on overlooked but prevalent contributors to multiple neurodegenerative pathologies. Glial dysfunction, vascular damage, metabolic dysregulation, and extracellular matrix breakdown appear across Alzheimer’s, Parkinson’s, vascular dementia, and ALS. They are tractable in concrete chemical and physical ways, unlike the cell-state and transcriptional failures of neurons themselves.
One such overlooked fulcrum is gamma oscillations. This is the rhythmic 40 Hz neural activity that helps to organize memory, attention, sleep, glymphatic flow, microglial function, and myelination. Gamma rhythms are profoundly disrupted early in Alzheimer’s. Restoring them with noninvasive 40 Hz audiovisual stimulation recruits microglia and clears amyloid in mouse models, with concurrent cognitive rescue (Iaccarino et al., Nature, 2016; Martorell et al., Cell, 2019).
The Spectris AD device. Image courtesy of Cognito.
Cognito Therapeutics is translating this into a therapy. Spectris AD is an at-home headset that delivers gamma stimulation noninvasively. Their Phase 2 OVERTURE preserved cognition, function, and brain volume out to 18 months (Hajós et al., Front Neurol, 2024; Cimenser et al., Front Syst Neurosci, 2021; Da et al., J Alzheimers Dis, 2024, Kern et al. Alzheimer’s Dement. 2025). The pivotal Phase 3 HOPE trial, which enrolled 673 patients across 70 U.S. sites, the largest device trial ever run in AD, reads out Q3 2026. We co-led the Series C.
When it comes to age-related vascular and lipid dysregulation, there are striking parallels between our two post-mitotic organs: heart and brain. For example, oxidized cholesterol species accumulate in aged cells and disrupt lysosomal function and autophagy pathways. This drives plaque formation in atherosclerosis, and in the aged brain it also manifests in microglia, the brain’s immune cells, which acquire a dysfunctional, proinflammatory state. Given that atherosclerosis is also one of the largest modifiable contributors to AD risk (Gottesman et al., JAMA Neurol, 2017), addressing oxidized lipids directly is a promising strategy for brain aging, an approach that Cyclarity Therapeutics is going at directly.
We’ve written extensively about the brain’s extracellular matrix elsewhere, which provides structural support for neurons and glia and interfaces with multiple pathways critical to brain aging. As we age, sugars in the body react with long-lived proteins to form permanent crosslinks called AGEs. The most abundant one in human tissue is glucosepane, and it’s a major reason blood vessels, skin, and cartilage get stiffer with age. Revel Pharmaceuticals engineers enzymes that cleave it.
Building the tools to unlock transformational therapies
The near-term bench treats patients, but it operates inside a brutal constraint: the preclinical models are wrong in kind, not just in detail. A rodent brain is a different organ from a human one: different cell types in different proportions, different microglial biology, different neuromodulatory systems, a different extracellular matrix, and a short lifespan. Worse, the field has spent decades arguing that compounds work in human cognition on the strength of mice navigating water mazes. Three chokepoints have kept the field from learning fast enough about aged brains: experimentation on whole human brain tissue, screening at scale, and the maps that connect brain biology to its function. We have supported efforts to address each.
The first chokepoint is human tissue. Drug discovery has run on cell lines and rodents because there has been no way to maintain cellular and molecular function in a whole human brain ex vivo. Bexorg changed that with BrainEx, a perfusion platform that restores molecular and cellular activity in postmortem human brains hours after death (Vrselja et al., Nature, 2019). Bexorg has now scaled to drug-development experiments on hundreds of donated human brains a year, many of them with the actual disease pathologies we need to treat. They are building XO Digital, a foundation model for human molecular neurobiology trained on the whole-brain data the platform generates.
The second chokepoint is screening at scale in tissue that has actually aged. Gordian Biotechnology closes that gap with pooled AAV screens in aged animals with thousands-fold improvements in throughput, the closest thing the field has to a systematic way to ask which targets still work in old tissue. The brain is a prime target tissue for the platform.
The third chokepoint is the maps. There is no detailed structural map of a mammalian brain, and there has not been one because the labor of manually verifying each reconstructed neural wire accounts for more than 90% of the cost of a whole-brain connectome. E11 Bio’s PRISM platform combines optical connectomics with molecular barcoding of individual cells across entire mammalian brains (E11 Bio PRISM, 2024), changing those economics. Knowing which connections exist, which cells make them, and how that architecture changes with age is prerequisite knowledge for precision therapies that act on specific circuits.
The other half of the third chokepoint is functional. Translating molecular and circuit-level changes into the lived experience of patients, what cognitive or psychiatric state corresponds to what underlying biology, is a prerequisite for any precision neurotherapeutic, and the mapping does not yet exist. Constellation is building a foundation model that does this directly, trained on real-time multimodal data: neural, physiological, behavioral, and environmental gathered at scales the field has not had access to before.
All of the above is upstream of the patient. Neurodegeneration is slow, and interventions are most valuable at the earliest deflection. Continuous wearable biomarker tracking developed by Adaptyx Biosciences enables passive monitoring; MaGIC Lifescience, a point-of-care multiplexed platform that reads pathogen, protein, and nucleic-acid biomarkers from blood, saliva, and urine in under twenty minutes, enables active diagnosis. They will create the sophisticated biomarkers uncovered by discovery platforms for effective translation and patient impact.
Pulling the curve forward
We have written that human minds, with their capacity for love, ambition, and creative work, are worth preserving. Philanthropists, investors, frontier-tech founders, and institutions like ARPA-H each have a part to play in advancing the brain-aging agenda. The work is to find the right shots and to build the tools that make the next round of shots better.
Changes in regulatory and economic regimes are critical to address aging: there is no FDA approval pathway for a drug that claims that patients age more slowly. Loyal is on track to be the first to create one, in dogs, initially. The path is the point: once aging is approvable as an indication in dogs, there is a template for approving one for humans, which could free a great deal of capital to work directly on brain aging.
Even modest interventions will return outsized value. As part of the Silver Linings project, our Longevity Policy lead Raiany Romanni modeled what pulling brain-aging interventions forward by even short intervals would mean for the United States: a one-year delay in brain aging adds an estimated $201B per year to U.S. GDP, with an $8.9T long-term return in net present value over decades. Investment today runs at a small fraction of what those returns justify, blocked by misaligned commercial incentives and short-sighted policy.
The patients who will benefit from the next decade’s brain-aging therapies are alive today, and their disease will not wait for those incentives to align. If you are a researcher, a founder, or a funder who wants to work on this, we want to hear from you. Reach us at info@amaranth.foundation.